- Blog
- Free coding bootcamps nyc
- Que es anime pdf
- Screaming bee voice changer free download
- Runemate switch between bots
- What is best app for video calling
- Epson scanner software cx9300 download
- Who sells nuance dragon software
- Brothers conflict passion pink english walkthrough
- Fm16 custom transfer database for apple
- Avast secureline vpn license key 2018
- Crystal reports download free trial
- Free mac disk partition software
- Alter ego game mark
- Epson scanner software big sur
- Allen bradley -dat files reader
- Tradeskillmaster tutorial
- Linux apache server tutorial 2016
- Arena mac eyeshadow
- Final fantasy ii snes versions
- Notepad for mac for html
- Jackson guitars made in japan
- Elvui update 7-2
- Nvidia nforce 430 drivers ubuntu
- Autodesk memento set scale and unit
- Most detailed printable periodic table of elements
- Os x lion iso mega
- Calligraphy software for windows 10
- Core keygen cs6 master collection mac
- Heroine full movie 720p free download
Valency is an important aspect of calculating chemical formulas for students and chemists. 6 This is the ability of an atom or a group of atoms to form chemical bonds with other atoms. Once you know the number of electrons of a chemical element, you can calculate its valency. All elements in the same period have the same number of electron shells but have different numbers of electrons and protons. In each period, the elements’ atomic numbers increase from left to right. The periodic table contains seven periods 5 (nine if you count the lanthanides and actinide series). The horizontal rows across the periodic table are called periods. Because atoms naturally occur with different numbers of neutrons-known as isotopes-the atomic mass is an average of all weights of all isotopes for a given atom. Under the element symbol is the atomic weight, which is the average weight of the protons and neutrons in an atom. 4 The atomic number, located at the top left of the element symbol, signifies the number of protons in an atom’s nucleus. How elements are organized on the periodic table:Įlements are organized in horizontal rows by increasing atomic number. 2 Oxygen (O) is the most common element in the Earth’s crust. Today, the 118 chemical elements identified on the periodic table include the materials that make up all known objects in the universe.ĭid you know? The most abundant element (by mass) making up Earth3 is Iron (Fe). He anticipated others would one day be discovered, so he left open spaces in his table for additions. Mendeleev’s periodic table included 63 elements.
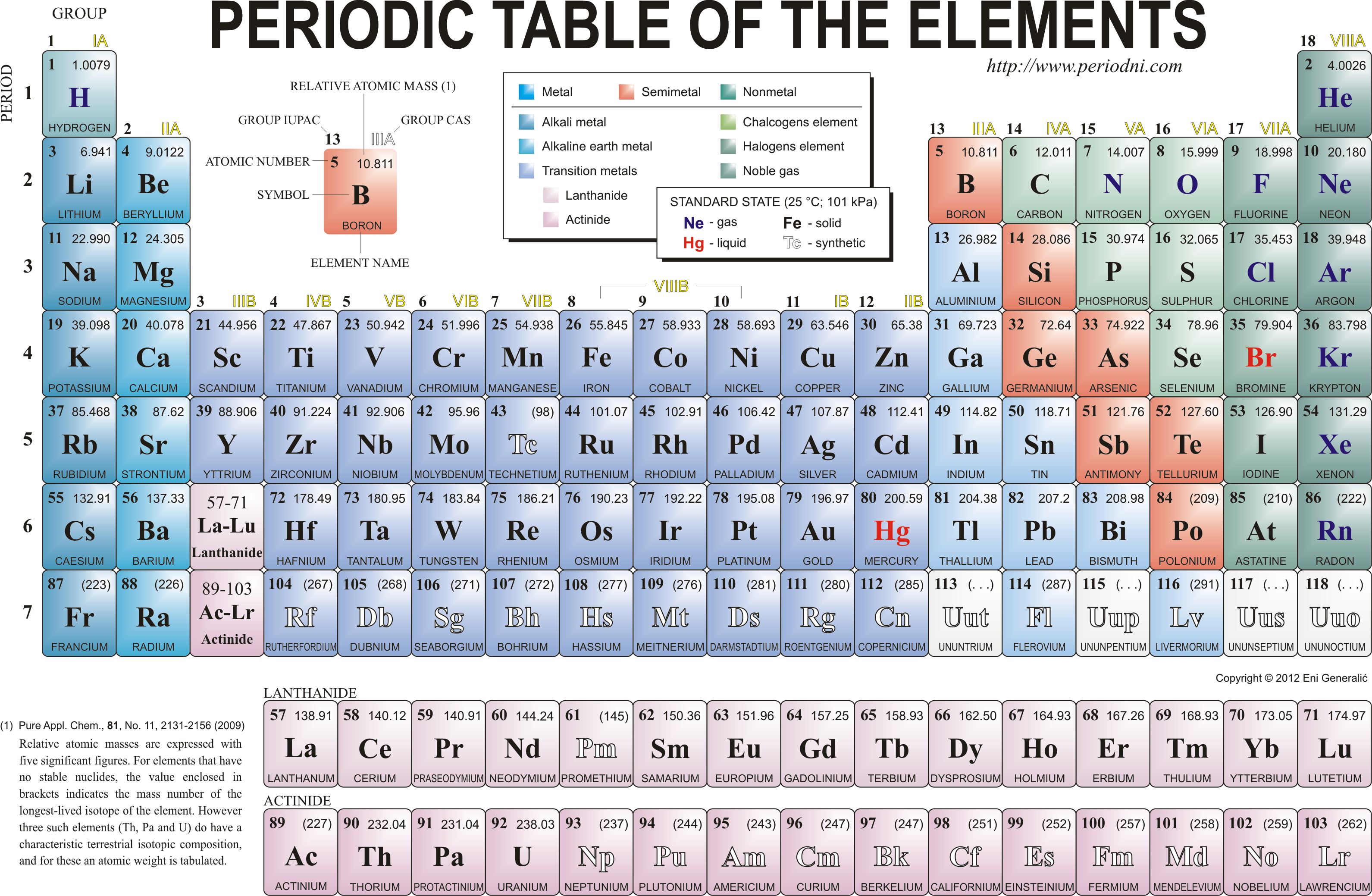
This structure helped Mendeleev and other scientists identify similarities and differences among elements to help predict future chemical reactions. He found a pattern, listing elements by their increasing atomic number and arranging them in a chart-creating the first periodic table. In 1869, Russian chemist Dimitri Mendeleev wanted to see if there was a pattern to the chemical properties of the elements he knew. You don’t have to be a chemist to appreciate the periodic table. These elements also create the products and technologies that have shaped modern societies in countless ways – clean drinking water, effective medicines and technologies ranging from solar panels to electronics components.

The chemical elements of the periodic table are the building blocks for everything in nature. These elements make up everything in our universe.
#MOST DETAILED PRINTABLE PERIODIC TABLE OF ELEMENTS PROFESSIONAL#
The periodic table on the Extensively illustrated and of use for those at school (via a special scholar version) to professional chemists.The Periodic Table of Elements 1 is an ordered system of all the identified elements. It has short summaries of important information about each element, up to element Number 111, including when first isolated, the origin of the name, what current uses or concerns are, and, rather curiously, the alchemical symbols. This Periodic Table is an on-line version of a fine poster produced by the Royal Society of Chemistry. Each element in the Chemicool has a link to a detailed description of that element.
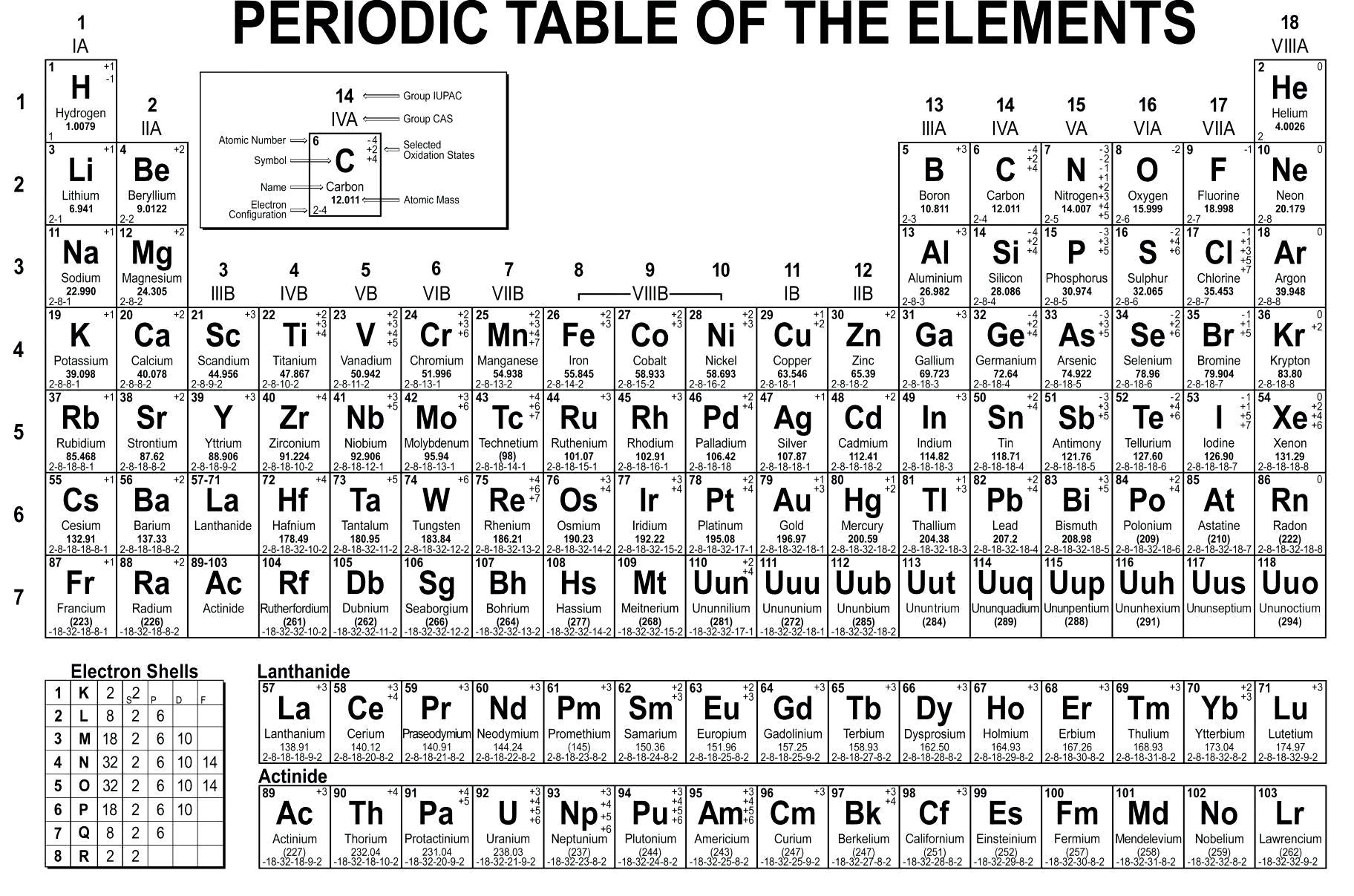
It contains detailed information about different elements of the periodic table. Entries include basic information, atomic structure, isotopes, facts, and related links.Ĭhemicool is an online comprehensive periodic table made by David Hsu of the Massachusetts Institute of Technology. An online, interactive periodic table created by Yinon Bentor.
- Blog
- Free coding bootcamps nyc
- Que es anime pdf
- Screaming bee voice changer free download
- Runemate switch between bots
- What is best app for video calling
- Epson scanner software cx9300 download
- Who sells nuance dragon software
- Brothers conflict passion pink english walkthrough
- Fm16 custom transfer database for apple
- Avast secureline vpn license key 2018
- Crystal reports download free trial
- Free mac disk partition software
- Alter ego game mark
- Epson scanner software big sur
- Allen bradley -dat files reader
- Tradeskillmaster tutorial
- Linux apache server tutorial 2016
- Arena mac eyeshadow
- Final fantasy ii snes versions
- Notepad for mac for html
- Jackson guitars made in japan
- Elvui update 7-2
- Nvidia nforce 430 drivers ubuntu
- Autodesk memento set scale and unit
- Most detailed printable periodic table of elements
- Os x lion iso mega
- Calligraphy software for windows 10
- Core keygen cs6 master collection mac
- Heroine full movie 720p free download
